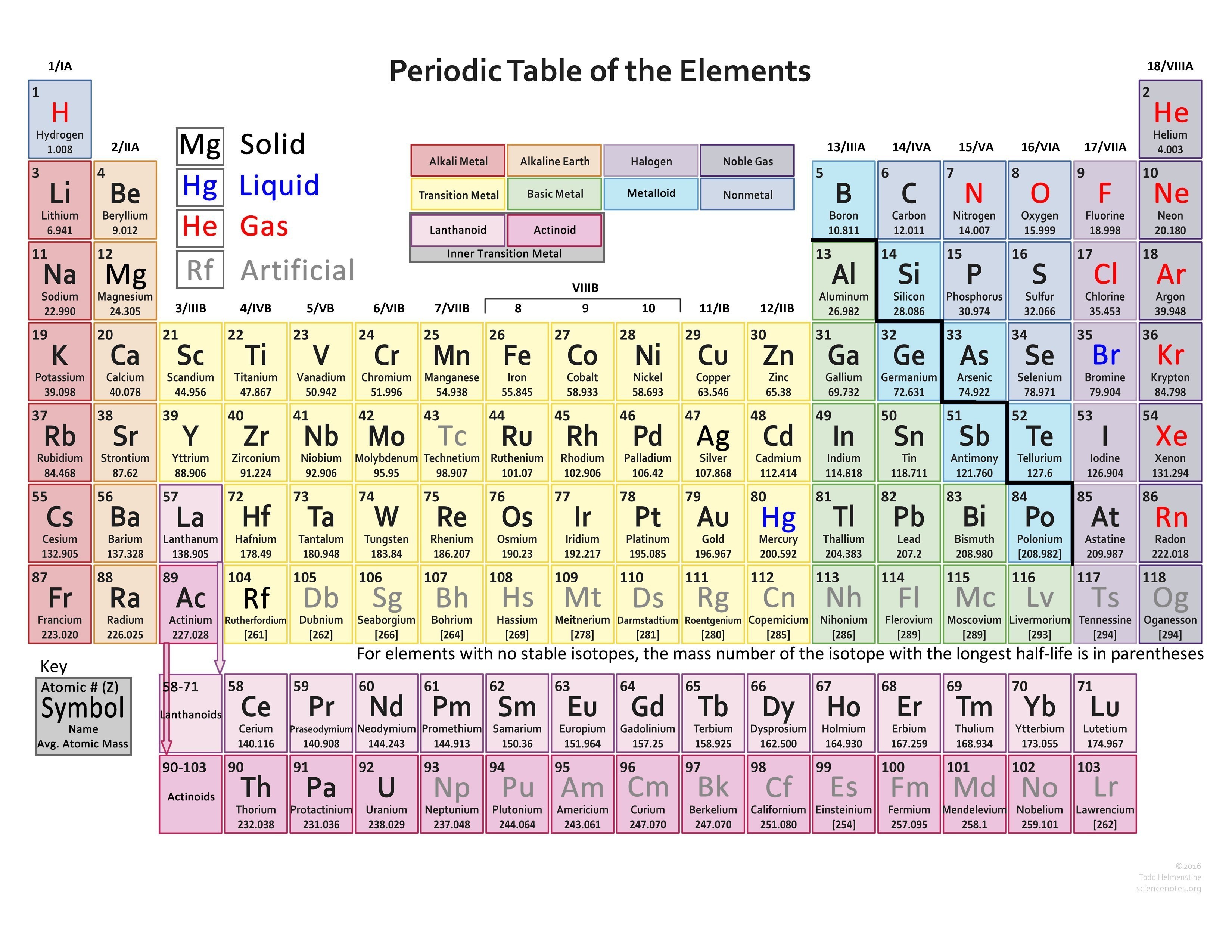
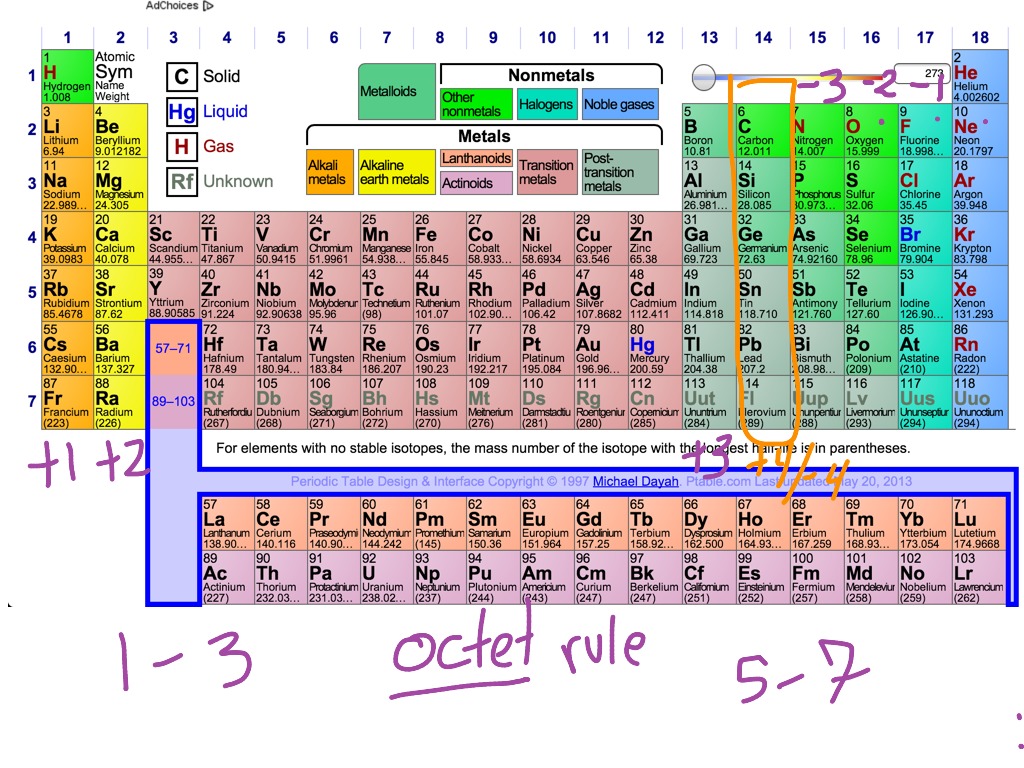
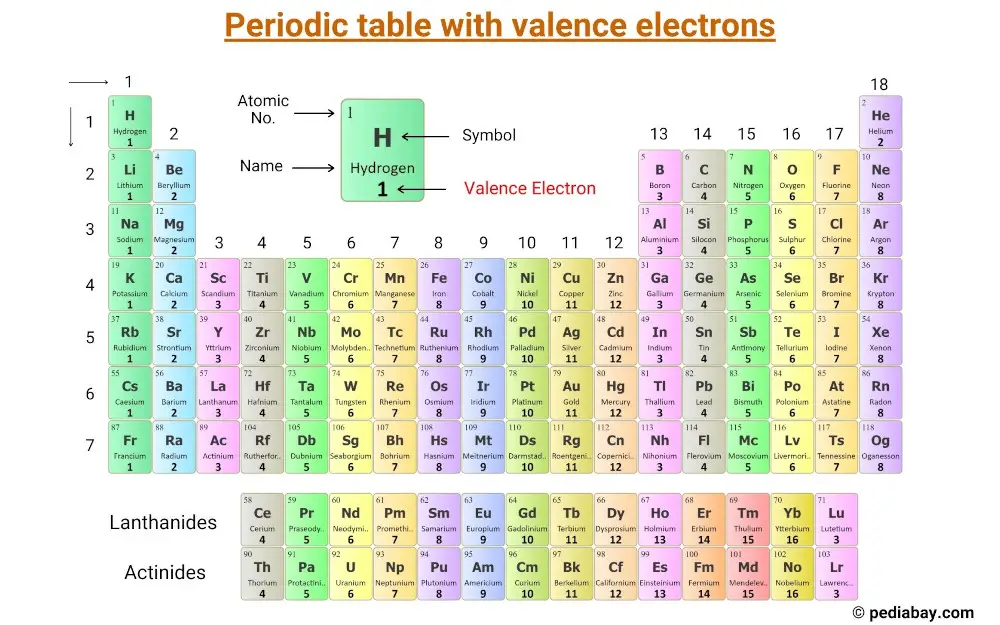
The electron configuration shows that the last shell of krypton has. The total number of electrons in a valence shell is called valence electrons. The last shell after the electron configuration is called the valence shell. The third step is to diagnose the valence shell. When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. Step-3: Determine the valence shell and calculate the total electrons. Again, principal shells with larger values of n lie at successively greater distances from the nucleus. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. The reason is the same as for atomic radii: shielding by filled inner shells produces little change in the effective nuclear charge felt by the outermost electrons.
Ionic radii follow the same vertical trend as atomic radii that is, for ions with the same charge, the ionic radius increases going down a column. Shannon, “Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides,” Acta Crystallographica 32, no. Valence electrons are the electrons present in the outermost shell of an atom. \): Ionic Radii (in Picometers) of the Most Common Ionic States of the s-, p-, and d-Block Elements.Gray circles indicate the sizes of the ions shown colored circles indicate the sizes of the neutral atoms.

